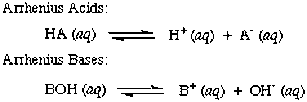District 7: Acids and Bases

http://sentryair.com/blog/wp-content/uploads/2012/10/Chemicals1.jpg
Welcome to your training for how to take down District 7 when you are in the arena. We all know the deadliness of acids and bases and it is important to remember this as you prepare to do battle with this District. The key is that you don't want to turn your back on this District for too long, otherwise they will likely kill you and then dispose of the body without leaving any evidence of the grotesque murder behind.
Past District 7 Champions
YOUR TRAINING BEGINS NOW
DIrections: Please take out your District 7 guided notes and review the following content:
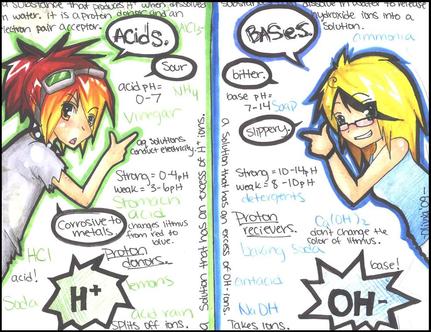
Properties of Acids and Bases
|
Use this powerpoint to study the properties.
When you dissolve an ionic compound in a solution it breaks apart into its ions. This positive and negative charge allows for the solution to now conduct electricity. You need charges to conduct electricity. Therefore, in order to conduct electricity you need ions. IONIC compounds conduct electricity and COVALENT compounds do not! |
2 Different Definitions of Acids and Bases *Honors*
Arrhenius Theory
Acids produce H+ when dissolved in a solution.
Bases produce OH- when dissolved in a solution. *Remember, H+ are hydrogen ions and OH- are hydroxide ions. |
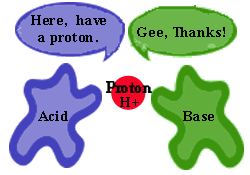
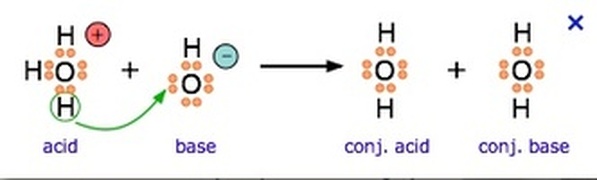
Bronsted-Lowry Theory
Acids donate H+ when dissolved in a solution.
Bases receive or accept the H+ when dissolved in solution. |
Identifying Acids and Bases in a Chemical Reaction
- Remember that acids and bases will always be listed as reactants if you are studying an acid/base reaction.
- Acids usually have a hydrogen ion. For example, HCl, HNO3, and H2SO4 all contain a hydrogen ion that can be donated to solution.
- Most bases contain an OH ion. Therefore, if a compound contains an OH, then it is a base. Bases are also compounds that can accept a hydrogen ion. For example, NH3 can accept a H in order to form NH4+. Therefore, the presence of OH OR the ability to accept a hydrogen is an indication that a compound is a base.
Strong versus Weak Acids/Bases
|
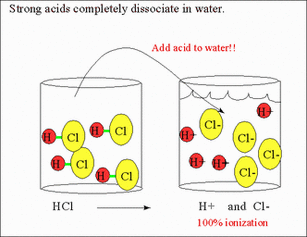
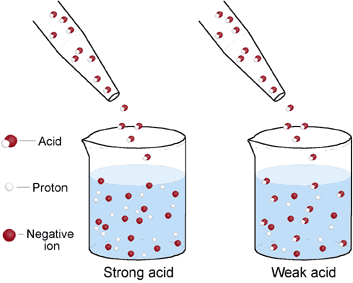
Acids/Bases can be classified as strong or weak based on how much it dissociates. When we say something disassociate this means to break apart. Acids/Bases that completely dissociate are called strong acids/bases. Acids/Bases that only partially dissociate are called weak acids/bases.
The seven strong acids are:
The seven strong bases are:
|
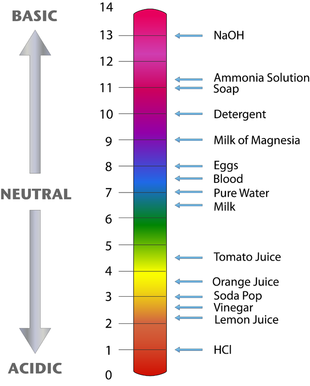
Measuring Acids and Bases: pH scale
pH + pOH = 14
|
The concentrations of acids and bases are often very low. We use the pH scale in order to more effectively convey the concentration of H+ in our solution. The pH scale is 0-14. An acid has a pH of 0-7 A base has a pH of 7-14 Neutral chemicals have a pH of 7.
*Honors* p(anything) means -log(anything), so pH means -log(H+). We calculate the pH by: pH=-log(H) We calculate pOH by: pOH=-log(OH) A strong acid/base means that the compound completely dissociates. This means that if we have a 0.1 M solution of HCl, then [H+]=0.1 M If we have a strong base, then that means that all of the compound dissociates. This means if we have a 0.1 M solution of NaOH, then we have [OH-]=0.1 M We can calculate the pOH if we have this. Once we have the pOH, we calculate the pH by: pH=14-pOH |
PRACTICE

Once you feel that you have reviewed the material, then try out these problems:
Answer Key:
Answer Key:
Need a sponsor
It is well known that having sponsors in the arena can be the difference between a win and a loss. Kill District 7 with a little flair and you are abound to get sponsors lining up! Take the assessment...