District 9: Reaction Rates and Equilibrium

Welcome to your training for how to take down District 9 when you are in the arena. This District breads truly ruthless killers. Not only are they ruthless, but they also kill extremely fast (it helps when you have a catalyst in your back pocket). They are advised by past champion Le Chatelier. Be very cautious when approaching this District, they have been known to make alliances with others and then kill them once the opportunity presented itself.
Past District 9 Champion
YOUR TRAINING BEGINS NOW
DIrections: Please take out your District 9 guided notes and review the following content:
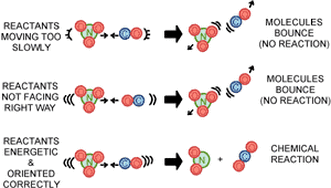
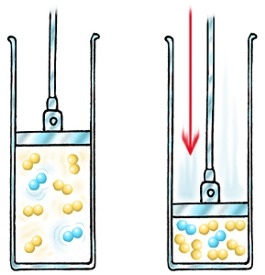
Factors affecting Reaction Rates
|
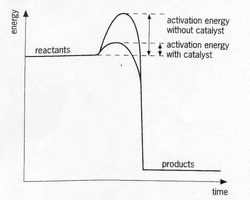
Activation Energy and Catalysts
|
Note: You can reuse catalysts! Catalysts do not get used up in a reaction.
|
Equilibrium
For the reaction in the video, think of heat as a product (on the RIGHT side).
- A reaction does not stop when equilibrium is achieved. The rate at which we use products/reactants is the same as the rate at which we produce products/reactions. We call this dynamic equilibrium where the reaction is still occurring, but equilibrium is maintained.
- Le Chatelier’s principle states that if we alter something that is on the products or reactants side of an equilibrium, then the system will respond in order to re-establish equilibrium.
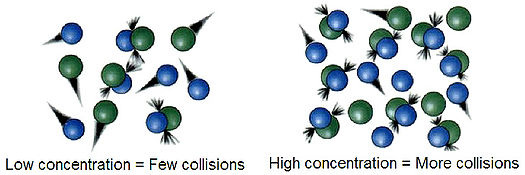
Concentration and Temperature
Temperature = heat, energy, kJ, J |
Pressure
|
PRACTICE

In order to take down this district, you will need a lot of practice! Complete the following practice problems:
Answer Key:
Answer Key:
Invitation to a feast...
The Capital is offering a "feast" at sunrise. District 9 is sure to be there. To ensure that you have the feast to yourself, complete the assessment to kill District 9...