District 8: Thermochemistry

Welcome to your training for how to take down District 8 when you are in the arena. This District possesses the power of fire, so there is no telling how they could harness these skills once they are let loose inside the Arena. This District is in it to win because they are one of only two Districts that has never won the Games. Knowing the skills of this District will be important if you hope to kill this District inside the arena. District 8 tributes are capable of killing you and telling you how many calories a kill took once the kill has been completed.
Past District 8 Champion
YOUR TRAINING BEGINS NOW
DIrections: Please take out your District 8 guided notes and review the following content:
Exothermic and Endothermic Reactions
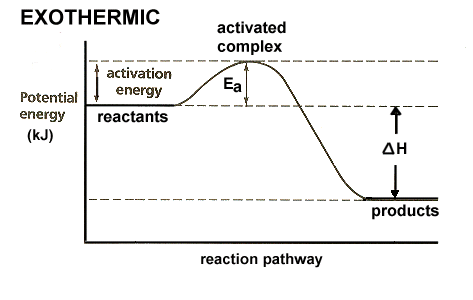
EXOTHERMIC
A reaction that releases energy during the reaction is known as an exothermic reaction. Energy is often given off in the form of heat.
Energy is listed as a product in the equation: Reactants --> Products + Energy |
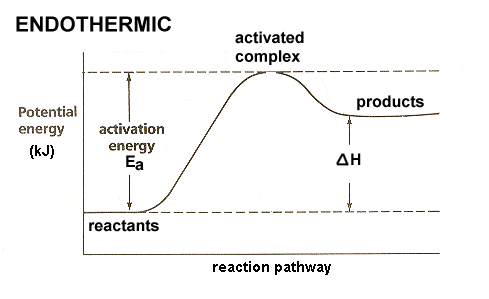
ENDOTHERMIC
A reaction that requires energy during the reaction is known as an endothermic reaction. Energy often is absorbed in the form of heat, so the substance feels cold.
Energy is listed as a reactant in the equation: Energy + Reactants --> Products |
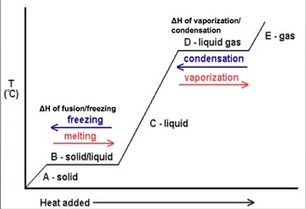
Phase changes can be classified as endothermic or exothermic. Since you have to add energy to go from a solid to a liquid and a liquid to a gas, these are listed as endothermic processes. Since you have to release energy to go from a gas to a liquid and a liquid to a gas, these are listed as exothermic processes.
*Honors*
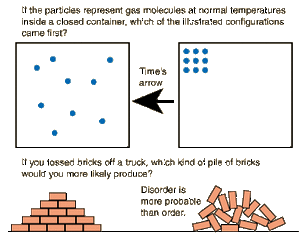
Entropy
|
Enthalpy
Free Gibbs Energy
We use free gibbs energy (G) to determine if a reaction is spontaneous or not spontaneous.
If G is negative (-), then the reaction is spontaneous.
If G is positive (+), then the reaction is not spontaneous. Remember, spontaneous means that the reaction will happen with no extra energy needed (besides activation energy). |
Skip "phase diagrams" on your guided notes.
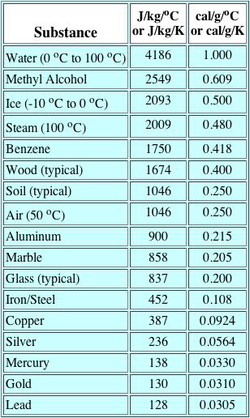
Specific Heat Capacity / Calorimetry
|
Heating Curve
Source: From-http://www.dlt.ncssm.edu/TIGER/Flash/phase/HeatingCurve.html
Areas of positive slope represent the solid, liquid or gas phases. Areas of no slope represent a phase transition.
Areas of positive slope represent the solid, liquid or gas phases. Areas of no slope represent a phase transition.
|
PRACTICE

Below you will find problems that will ensure a fast kill of the District 8 tributes:
Answer Key:
Answer Key:
Fire!!! Aghhhhh
The District 8 tributes are masters of fire, so the woods behind you being on fire is likely their doing. As you run, you feel knives being thrown at your back by the fire wielding District 8 tribute. Complete the assessment so that you can fight back against them!