District 2: Periodic Table and Trends
http://www.ptable.com/Images/periodic%20table.png
Welcome to your training for how to take down District 2 when you are in the arena. Where would these other Districts, and the Capital for that matter, be without the Periodic Table? District 2 has a deep understanding of elements and how they work, so you are going to need to become an expert if you are going to take this District down. They are advised by past champion, Dmitri Mendeleev. It is rumored that he has gone crazy and this may be useful information once you are inside the arena.
Past District 2 Champion
YOUR TRAINING BEGINS NOW
Your training resources (powerpoints)
DIrections: Please take out your District 2 guided notes and review the following content:
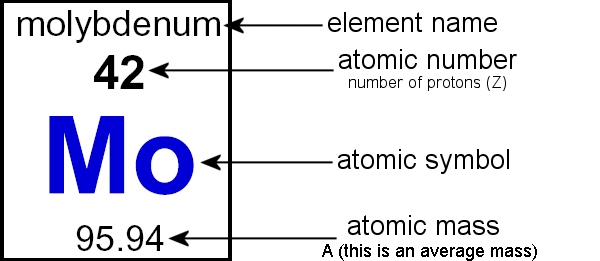
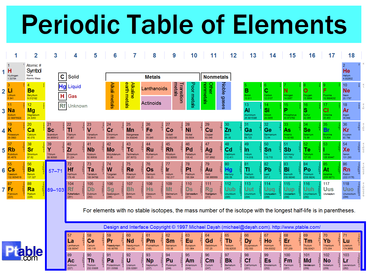
The Periodic TableAn element is a substance that cannot be separated/broken down into simpler substances by chemical means. An element consists of only one type of atom
On the Periodic Table, we are told the following about every element:
|
http://www.youtube.com/watch?feature=player_embedded&v=TIxiDESxc0I
|
The Many Families of the Periodic Table
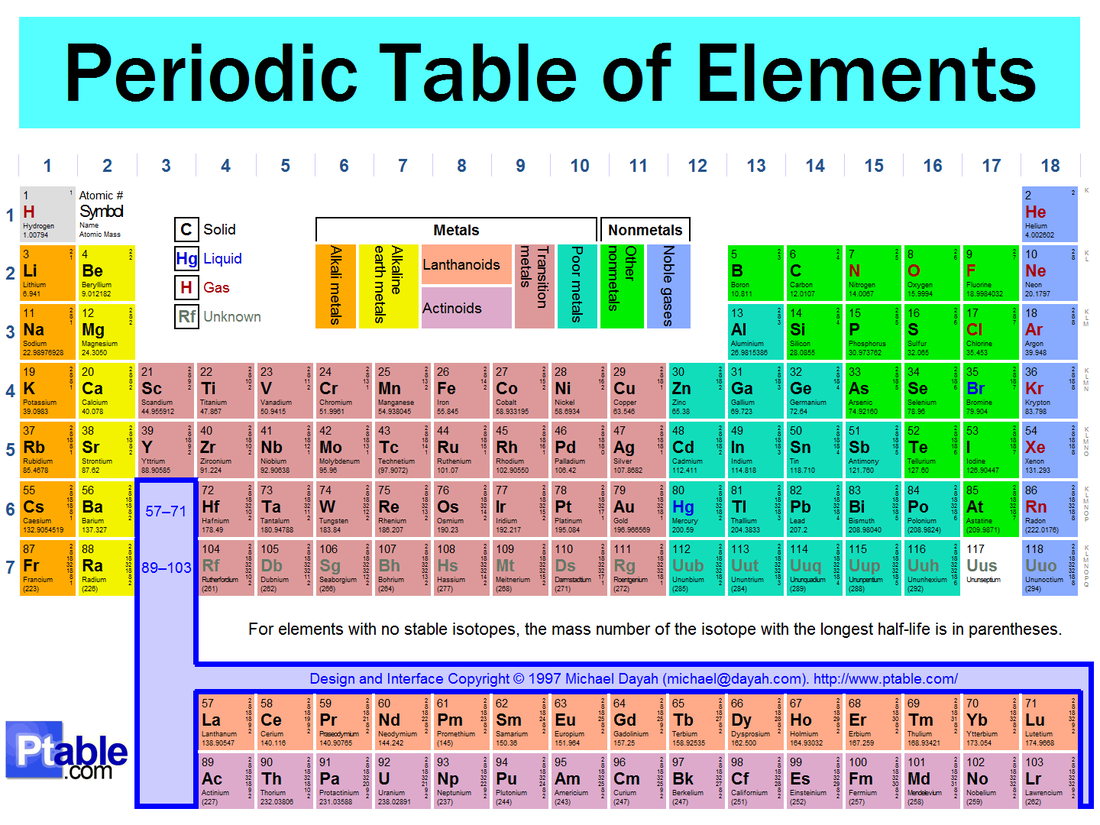
Groups or FamilesA group is a vertical column on the Periodic Table. Elements in the same group have similar properties because they all have the same number of valence electrons. Therefore, each group on the Periodic Table has special names and properties.
|
PeriodsA period is a horizontal row on the Periodic Table. The period can tell you the number of energy levels an atom or ion has.
|
Alkali metals- Group 1A or 1
- 1 valence electron - Highly reactive with water. - Cannot find these elements on their own in the world, they are always bound to other elements. |
Alkaline Earth metals- Group 2A or 2
- 2 valence electrons - Highly reactive, but not as reactive as Alkali Metals |
Transition metals- Groups 3 - 12
- typical properties of metals (conductor, malleable, etc.) |
Halogens- Group 7A or 17
- 7 valence electrons - This is the most reactive group on the Periodic Table because it only needs to get 1 electron to complete the Octet Rule. |
Noble Gases- Group 8A or 18
- 8 valence electrons - Non-reactive - It already has a full octet, so there is no need to react because it has all the electrons that it needs. |
METALS, NONMETALS, AND SEMIMETALS
|
The “staircase” separates metals from non-metals on the Periodic Table. Elements to the left of the staircase are metals. Elements to the right of the staircase are non-metals. Elements that touch the staircase (except aluminum) are semimetals.
There is one exception to the rule and that is hydrogen. Hydrogen is located to the left of the staircase, but hydrogen is a non-metal. |
PERIODIC TRENDS
http://www.youtube.com/watch?feature=player_embedded&v=HmvpXdxAZrc
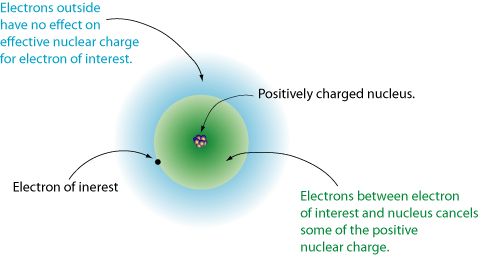
IONIZATION ENERGYThe negatively charged electrons are attracted towards the positively charged nucleus. This nuclear attraction plays a major role in the periodic trends that are observed for elements.
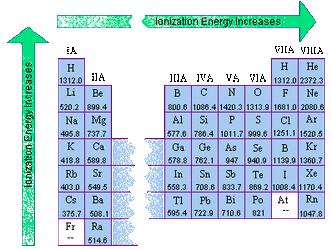
Ionization energy is the energy that is required to remove an electron from an atom. This is the energy that is required to turn our neutral atom into an ion. This is different depending on the element. How nuclear attraction affects ionization energy: As you go down a group more electron orbits are added. Ionization energy decreases as you go down a group because it requires much less energy to remove a valence electron due to the shielding. As you move across a period you add more protons and electrons within the same electron orbit. The larger amount of protons in the nucleus and the larger amount of electrons show an increased attraction. It is much harder to pull away electrons as you add more protons within a period, so ionization energy increases. |
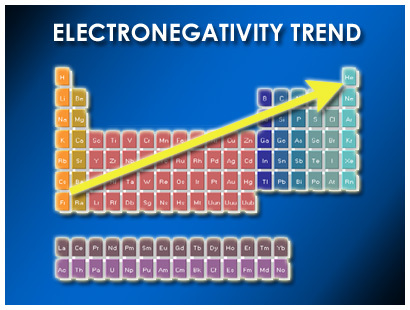
ELECTRONEGATIVITYElectronegativity is the measure of the ability of an atom to attract electrons. We can also call this the strength of the atom.
As you go down a group, the electronegativity of an element decreases. As you go across a period, the electronegativity of an element increases. How nuclear attraction affects electronegativity: As you go down a group more electron orbits are added. Electronegativity decreases because there is a decreased ability of the positively charged nucleus to attract valence electrons as a result of this larger distance. As you move across a period you add more protons and electrons within the same electron orbit. The larger amount of protons in the nucleus and the larger amount of electrons show an increased attraction. This results in a much larger ability for the positively charged nucleus to attract valence electrons. |
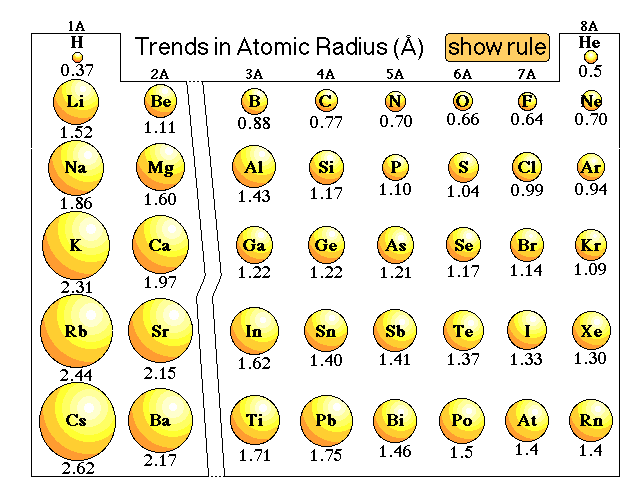
Atomic RadiusThe atomic radius is the distance from the center of the nucleus to the outermost edge of the electron cloud for an atom.
How nuclear attraction affects atomic radius: As you add more electron shells to an element, the element becomes “bulkier” due to the increased amount of electron orbits. This means that as you go down a group, more orbits are added, so the atomic radius becomes bigger. As you move across a period, the atomic radius decreases. More protons are added to the nucleus and more electrons are added within the same electron orbit. This means there is a larger positive and negative charge, which results in a higher attraction. |
PRACTICE!

http://cdn.thedeadbolt.com/wp-content/uploads/2012/03/the-hunger-games-training.jpg
The Mysterious Elements
The District 2 tributes have a large amount of elements at their disposal, but you were able to obtain a vile of mercury at the cornucopia. You dip your arrow into the vile and aim it at the leg of the tributes (mercury poisoning is sure to get you sponsors). To see if this is successful, complete the following assessment...