District 5: Stoichiometry and Solutions

http://sentryair.com/blog/wp-content/uploads/2012/10/Chemicals1.jpg
The Capitol is so infatuated with stoichiometry and solutions that they granted them two Districts (as opposed to one that all other Districts are allotted). Additionally, the Capitol showers them with praise throughout the year by providing them with more food and supplies then all the other Districts. This District is just as fierce and destructive as their sister District (District 4). Many Hunger Games have come down to a battle royale between District 4 and 5. You will need to have a profound understanding of stoichiometry and solutions in order to take this District down because they possess the power to perform the ultimate kill move: the grams to moles to moles to grams conversion.
YOUR TRAINING BEGINS NOW
DIrections: Please take out your District 5 guided notes and review the following content:
Intermolecular Forces
|
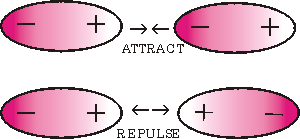
Intermolecular forces are forces that attract molecules to one another. The three main types of intermolecular forces are:
|
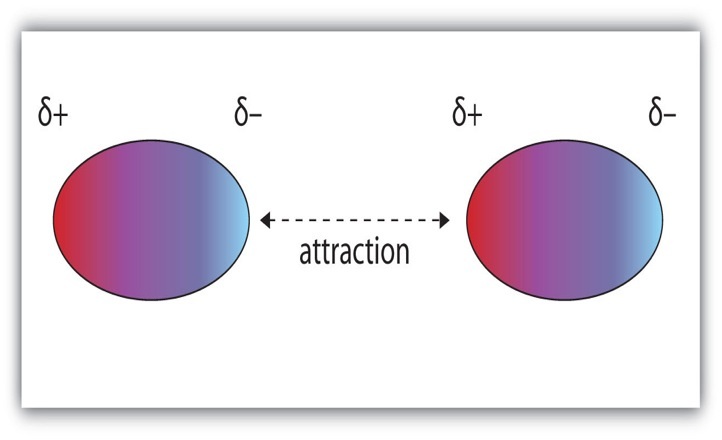
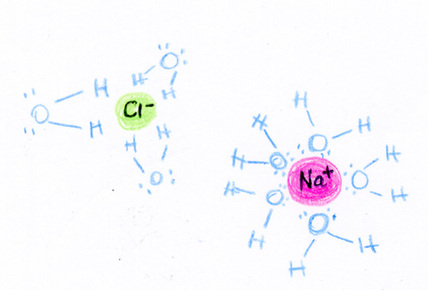
Dipole-Dipole ForcesDipole-dipole forces refer to the attraction between molecules that have a permanent dipole. A dipole is when one side is positive and the other side has negative charge. The positive end of one molecule will be attracted to the negative end of the other molecule.
|
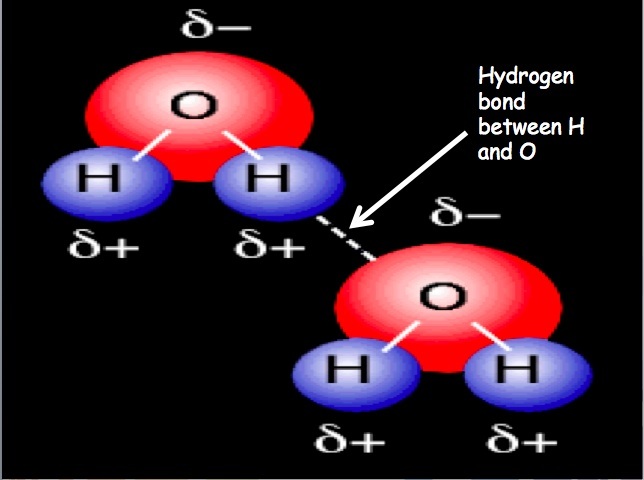
Hydrogen BondingThe attraction of one molecule that contains hydrogen to another molecule. When hydrogen is bound to N, O, or F, there is a dipole. The partial positive charge on hydrogen is attracted to N, O, or F of another molecule. This results in a hydrogen bond.
|
Dispersion/Van der Waals ForcesSome molecules do not have dipoles within the molecule (e.g. O2), but they can have an induced dipole. Dispersion forces are the intermolecular forces resulting from the uneven distribution of electrons and the creation of temporary dipoles. This is the weakest intermolecular force.
|
What affects solubility??
In order to dissolve a solute in a solvent, the solvent must use intermolecular forces in order to break the solute apart and pull it into solution.
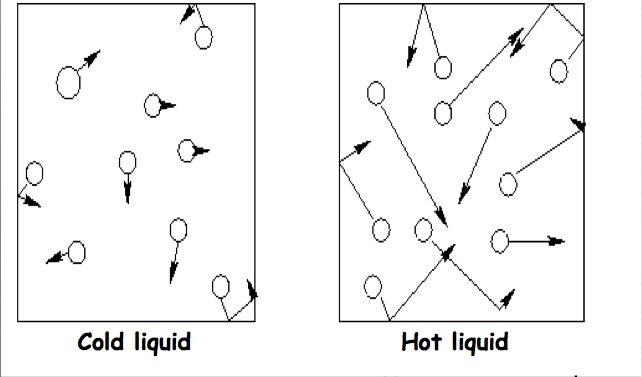
Change TEMPERATURE?As you increase the temperature, the rate at which the solute dissolves increases.
As you increase the temperature, molecules start moving faster. By moving faster, the molecules are better able to grab the solute, form the cages around them and pull them into the solution. Therefore, the faster molecules are moving, the faster something dissolves. |
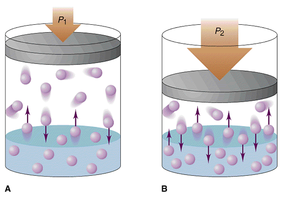
Change PRESSURE?Pressure only affects how GASES dissolve!
When pressure increases, it pushes the gases to dissolve into the solvent! As pressure increases, the dissolution rate increases. |
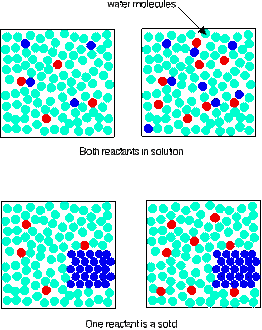
Change SURFACE AREA?High surface area = break into small pieces
Low surface area = 1 big piece Increase surface area to expose the solute to the solvent. As surface area increases, the dissolution rate increases. << http://www.chemguide.co.uk/physical/basicrates/conceffect.gif |
The big kill move: going from grams to grams
Watch the video above to see an example of a stoichiometry question where you will use grams of product to calculate the grams of reactant needed.
PRACTICE

- PracticeAnswer Keys:
Get that arrow ready...
While hiding out in the tree you spot the District 5 tribute walking below. You pull your arrow out, aim, and to see if you kill this District, then complete the assessment...