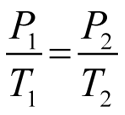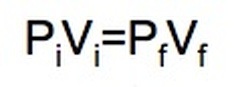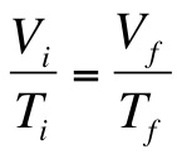District 6: Gases

http://sentryair.com/blog/wp-content/uploads/2012/10/Chemicals1.jpg
Welcome to your training for how to take down District 6 when you are in the arena. The gases are one of the major players in these Games. The gas laws are central to so many things that occur throughout the universe and this District has a very intimate knowledge of how gases can be manipulated for their own advantage. With the advisers Boyle, Gay-Lussac, and Charles, you can expect that this District will go far during the Games. The ability to crush a human being without ever touching them is one of their signature kill moves.
Past District 6 Champions
YOUR TRAINING BEGINS NOW
DIrections: Please take out your District 6 guided notes and review the following content:
Pressure
|
Diffusion
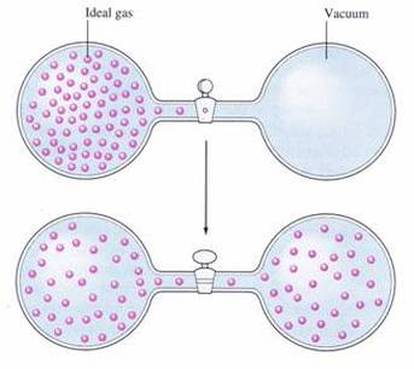
Diffusion is the movement of particles from regions of higher density to regions of lower density. Because gases are constantly moving in random motion, they spread out from one another very quickly. The random motion of gases is what explains the process of diffusion and the rate at which gases diffuse. Gases move very fast. Therefore, gases diffuse through a room at a relatively fast rate.
Absolute Zero and the Kelvin Scale
|
To convert from Celsius to Kelvin, we use: Kelvin = Celsius + 273 To convert from Kelvin to Celsius, we use: Celsius = Kelvin – 273 |
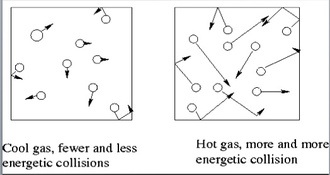
The temperature that we feel is created by the movement of atoms. The movement of gas molecules in the air creates the observable temperature that we experience each day.
The 2 main temperature scales in chemistry are the Celsius and Kelvin scales.
|
GAS LAWS
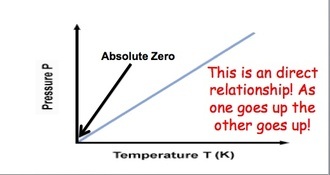
GAY-LUSSAC'S LAWFocus on PRESSURE and TEMPERATURE.
Gas molecules move quicker the more we heat them up. This means that they will be hitting the sides of the container more often and the pressure will also increase as the temperature increases. If we keep volume constant, then pressure and temperature are directly related. |
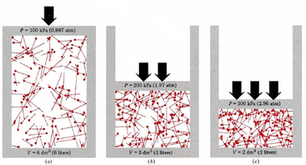
BOYLE'S LAW
|
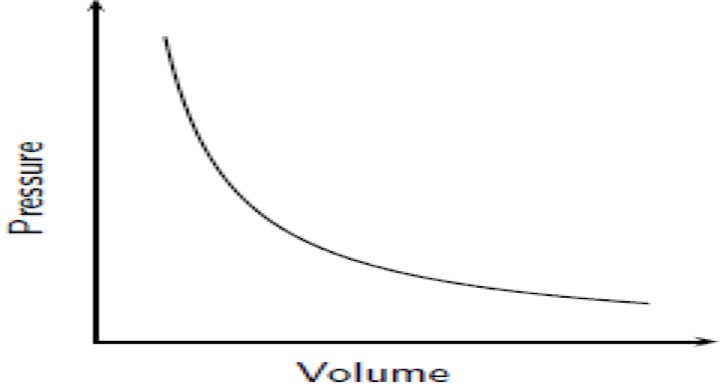
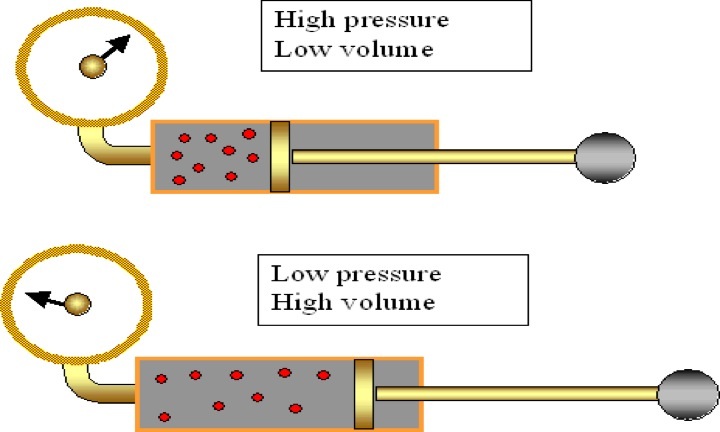
Focus on PRESSURE and VOLUME.
As pressure goes up, the volume goes down. If the container gets larger the gas molecules are hitting the walls at a less frequent rate. Therefore, by increasing the volume of a container, we are decreasing the pressure that is felt by that container. See picture >> |
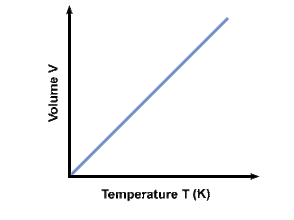
CHARLES' LAW
|
Focus on VOLUME and TEMPERATURE.
As you heat up gas molecules, they move quicker and collide with the surfaces of the container more. If the container is capable of expanding, so that pressure can remain constant, then the volume will increase as the temperature increases. Charles Law can be described by the equation: |
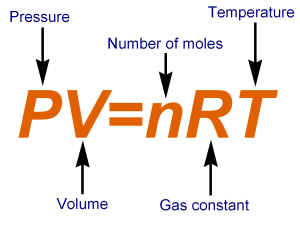
IDEAL GAS LAW *HONORS*
|
STP:
Scientists use the idea of standard temperature and pressure (STP) to describe gases. Scientists have designated STP as being 1 atm and 0 C or 273 K. Many gas law problems use STP. The ideal gas law combines Boyle’s, Charles , and Gay-Lussac’s Law into one single equation. An ideal gas is an imaginary gas whose particles are infinitely small and do not interact with each other. The law works best at room temperature and atmospheric pressure. The value of R is on the back of your CST periodic table. |
PRACTICE

When you feel that you have covered an adequate amount of material in this section, then try the following problems:
Answer Keys:
Answer Keys:
Watch out for those tracker jackers!
The Gamemakers have become board with the action in the arena, so they release tracker jackers on you. While running away from them you run into the District 6 tribute. To determine whether you kill them, complete the assessment...