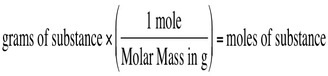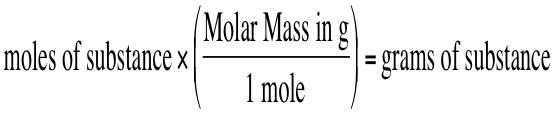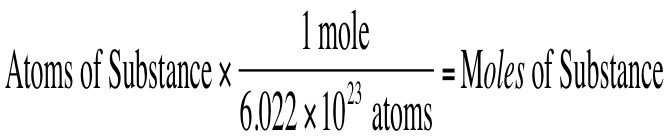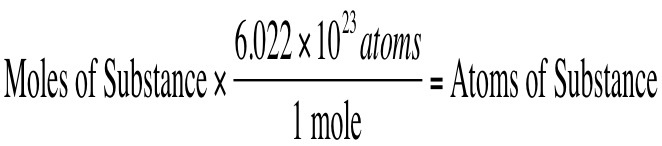District 4: Stoichiometry and Solutions

This is one of two powerhouse Districts. There is rarely a Hunger Games that does not have these tributes in the final group of individuals fighting to the death. The mere utterance of their name is enough to strike fear into the hearts of their opponents. If there is going to be a District that is going to take you down, this may be the one that does it. It is for this reason that intense training will be necessary if you are to ensure that you are victorious! They have many weaknesses, but these weaknesses can only be exploited if you have adequate training.
YOUR TRAINING BEGINS NOW
Your training resources (powerpoints)
DIrections: Please take out your District 4 guided notes and review the following content:
CHEMICAL EQUATIONS
|
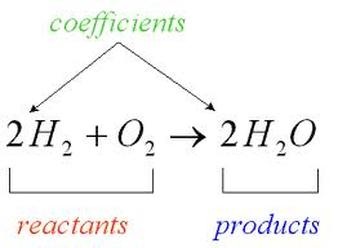
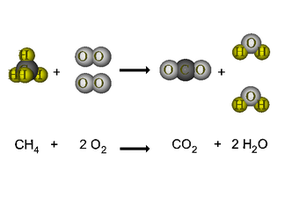
The law of conservation of mass explains that matter cannot be created or destroyed, it can only change forms.
In a chemical reaction, we have reactants and products. Reactants --> Products A molecule can be in a solid, liquid, gas, or aqueous state.
|
BALANCING CHEMICAL EQUATIONS
Watch the first video at this website.
We CANNOT change the subscripts. The only thing we can change is the number in front of the element/compound. This is called a coefficient.
still struggling? check this video out... ONLY WATCH AS MUCH AS YOU NEED, you have more training to do!
http://www.youtube.com/watch?v=_B735turDoM
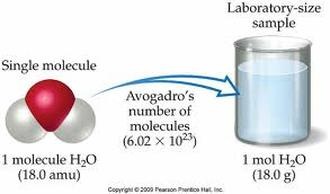
The MOLE
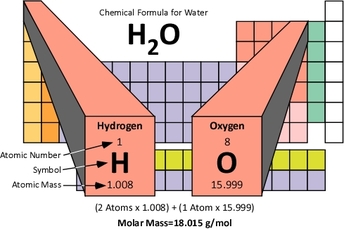
If we have 1 MOLE of something, how heavy it is is called MOLAR MASS.1. How many atoms of each element are there?
**If there are parentheses, multiply the inside number by the outside number. (Ex: Ca(OH)2 will have 2 O and 2 H.) 2. What is the atomic mass for each element? 3. Multiply! 4. Add! 1 mole of H2O is 18 grams! That's how heavy 6.02x10^23 molecules of water is. |
Find grams from moles OR Find moles from grams
Think!
- What do you start with?
- What goes on the bottom of the next fraction?
- What goes on top?
- Can you cancel? Then multiply and divide!
Find particles from moles OR find moles from particles
PARTICLES = ATOMS, MOLECULES, COMPOUNDS, FORMULA UNITS
Think!
Think!
- What do you start with?
- What goes on the bottom of the next fraction?
- What goes on top?
- Can you cancel? Then multiply and divide!
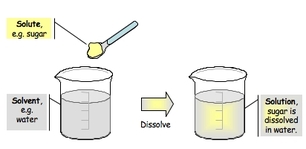
Solute and Solvent
|
http://www.youtube.com/watch?v=3G472AA3SEs
|
When studying solutions and how to make them, there are two primary ingredients that exist in every chemical solution: the solute and the solvent.
When water is the solvent, it is an aqueous solution (which we talked about above). |
Measuring the concentration ("how strong") a solution is:
|
http://www.youtube.com/watch?v=8oTqwBAvbnY
|
Follow the equation on the left to find "molarity" of a solution.
Practice Problem: What is the molarity of 245.0 g of H2SO4 dissolved in 1.00 L of solution? Highlight for answer: x = 2.498 M What is the molarity of 5.30 g of Na2CO3 dissolved in 400.0 mL solution? Highlight for answer: x = 0.125 M |
PRACTICE

- 4.1 - Balancing Equations Practice
- 4.2 - Molar Mass and Grams to Moles
- 4.3 - Molarity
- 4.4 - Moles to Particles
ANSWER KEY
A fight for your life...
You emerge from the forest and you discover the District 4 tribute waiting with an ax ready to take a swing at your head. Complete the assessment in order to kill this District...